Search Results
Showing results 1 to 20 of 63

A Funny Taste
Source Institutions
In this activity, learners explore the different salinities of various sources of water by taste-testing.

Salt 'n Lighter
Source Institutions
In this activity, learners discover that as the salinity of water increases, the density increases as well. Learners prove this by attempting to float fresh eggs in saltwater and freshwater.

Diet Light
Source Institutions
In this quick activity, learners observe how the added sugar in a can of soda affects its density and thus, its ability to float in water.

Underwater Fireworks
Source Institutions
In this activity, learners investigate diffusion by creating underwater "fireworks" using food coloring, oil and water.

Dry Ice Comet
Source Institutions
In this activity, dry ice and other items are used to construct a demonstration model of a comet that illustrates the comet nucleus, coma, and tails.

Why Doesn’t the Ocean Freeze?
Source Institutions
In this activity, learners explore how salt water freezes in comparison to fresh water.

Introduction to the New Chain Gang
Source Institutions
In this activity, learners use pop-beads to understand the characteristics and properties of polymer chains.

It's a Gas, Man
Source Institutions
In this activity, learners discover if carbon dioxide has an effect on temperature.

Rubber Band Thermodynamics
Source Institutions
In this demonstration, learners explore the thermal properties of rubber. Learners investigate whether a rubber band contracts or expands when heated.

Create Gas
Source Institutions
Learners mix vinegar and baking soda together in a bottle to create a chemical reaction. The reaction produces a gas, carbon dioxide, which inflates a balloon attached to the mouth of the bottle.

Lava Lamps
Source Institutions
Learners observe working lava lamps to understand how they work (included in PDF link).

Imploding Pop Can
Source Institutions
In this dramatic activity/demonstration about phase change and condensation, learners place an aluminum can filled with about two tablespoons of water on a stove burner.

Crunch Time
Source Institutions
In this quick and easy activity and/or demonstration, learners use two empty 2-liter bottles and hot tap water to illustrate the effect of heat on pressure.

Do Cities Affect the Weather?
Source Institutions
In this activity, learners explore clouds and how they form.

Can You Make Ice Cream in Two Minutes?
Source Institutions
In this demonstration, learners observe how liquid nitrogen both boils and freezes ingredients to make ice cream in two minutes.
Finding the Right Crater
Source Institutions
This quick demonstration (on page 11 of PDF) allows learners to understand why scientists think water ice could remain frozen in always-dark craters at the poles of the Moon.
Pepper Scatter
Source Institutions
In this activity, learners explore the forces at work in water. Learners experiment to find out what happens to pepper in water when they touch it with bar soap and liquid detergent.
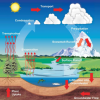
Weather Stations: Phase Change
Source Institutions
In this activity, learners observe the water cycle in action! Water vapor in a tumbler condenses on chilled aluminum foil — producing the liquid form of water familiar to us as rain and dew.

Toasty Wind
Source Institutions
In this quick activity, learners use a toaster to investigate the source for the Earth's wind. Learners hold a pinwheel above a toaster to discover that rising heat causes wind.
Floating Candles
Source Institutions
In this chemistry activity, learners observe a combustion reaction and deduce the components necessary for the reaction to occur.
