Search Results
Showing results 1 to 20 of 69

Matter on the Move
Source Institutions
Learners observe and conduct experiments demonstrating the different properties of hot and cold materials.

Skin Deep
Source Institutions
In this activity, learners explore how to protect their skin while applying pesticides to plants.

A Funny Taste
Source Institutions
In this activity, learners explore the different salinities of various sources of water by taste-testing.

Salt 'n Lighter
Source Institutions
In this activity, learners discover that as the salinity of water increases, the density increases as well. Learners prove this by attempting to float fresh eggs in saltwater and freshwater.

Diet Light
Source Institutions
In this quick activity, learners observe how the added sugar in a can of soda affects its density and thus, its ability to float in water.

Underwater Fireworks
Source Institutions
In this activity, learners investigate diffusion by creating underwater "fireworks" using food coloring, oil and water.

Dunking the Planets
Source Institutions
In this demonstration, learners compare the relative sizes and masses of scale models of the planets as represented by fruits and other foods.

Why Doesn’t the Ocean Freeze?
Source Institutions
In this activity, learners explore how salt water freezes in comparison to fresh water.

Evaporation
Source Institutions
This three-part activity consists of an activity that groups of learners develop themselves, a given procedure, and an optional demonstration.

It's a Gas, Man
Source Institutions
In this activity, learners discover if carbon dioxide has an effect on temperature.

Beating Gravity
Source Institutions
In this demonstration, learners watch as a device drops a ball into a cup without touching the ball or cup, even though the ball and cup are virtually side-by-side.

Forgotten Genius
Source Institutions
This series of chemistry stations is designed to accompany the PBS documentary about African-American chemist "Percy Julian: Forgotten Genius." Each of the six stations features either a chemical or p

Imploding Pop Can
Source Institutions
In this dramatic activity/demonstration about phase change and condensation, learners place an aluminum can filled with about two tablespoons of water on a stove burner.

Crunch Time
Source Institutions
In this quick and easy activity and/or demonstration, learners use two empty 2-liter bottles and hot tap water to illustrate the effect of heat on pressure.

Do Cities Affect the Weather?
Source Institutions
In this activity, learners explore clouds and how they form.

Can You Make Ice Cream in Two Minutes?
Source Institutions
In this demonstration, learners observe how liquid nitrogen both boils and freezes ingredients to make ice cream in two minutes.

Egg-Citing Physics
Source Institutions
In this demonstration about momentum, use physics to distinguish between a hard-boiled egg and a raw egg without cracking them open.
Pepper Scatter
Source Institutions
In this activity, learners explore the forces at work in water. Learners experiment to find out what happens to pepper in water when they touch it with bar soap and liquid detergent.
Exploring the Ocean with Robots
Source Institutions
In this activity, learners are introduced to robotic submarines called gliders. Learners make “gliders” from plastic syringes and compare these to Cartesian bottles and plastic bubbles.
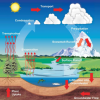
Weather Stations: Phase Change
Source Institutions
In this activity, learners observe the water cycle in action! Water vapor in a tumbler condenses on chilled aluminum foil — producing the liquid form of water familiar to us as rain and dew.
