Search Results
Showing results 1 to 20 of 64

A Funny Taste
Source Institutions
In this activity, learners explore the different salinities of various sources of water by taste-testing.

Exploring Materials: Liquid Crystals
Source Institutions
In this activity, learners discover that the way a material behaves on the macroscale is affected by its structure on the nanoscale.
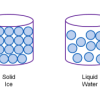
Atoms and Matter (3-6)
Source Institutions
In this activity, learners build models of atoms and molecules, then consider their role in different phases of matter, density, and mixtures and solutions.

Sublime Sublimation
Source Institutions
In this activity, learners explore sublimation by conducting experiments with dry ice.

Odors Aloft
Source Institutions
Learners smell balloons filled with different scents to guess what's inside. From this, they infer the presence and motion of scented molecules.

Dunking the Planets
Source Institutions
In this demonstration, learners compare the relative sizes and masses of scale models of the planets as represented by fruits and other foods.
Heating and Cooling of the Earth's Surface
Source Institutions
Learners conduct an experiment to determine the rate at which two materials, sand and water, heat up and cool down.
Twirling in the Breeze
Source Institutions
In this engineering activity, learners build a device (an anemometer) to measure how fast the wind is blowing.

Forgotten Genius
Source Institutions
This series of chemistry stations is designed to accompany the PBS documentary about African-American chemist "Percy Julian: Forgotten Genius." Each of the six stations features either a chemical or p

Submarine: Soda Cup Lander
Source Institutions
In this activity (on page 2), learners create a submarine using a plastic cup. This is a fun way to learn about buoyancy and density.
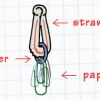
Deep Sea Diver
Source Institutions
In this ocean engineering activity, learners explore buoyancy and water displacement. Then, learners design models of deep sea divers that are neutrally buoyant.

Oily Ice
Source Institutions
In this activity, learners experiment with the density of ice, water, and oil. Learners will discover that the density of a liquid determines whether it will float above or sink below another liquid.

Sail Away
Source Institutions
In this activity, learners explore watercraft engineering and sailing.
Finding the Right Crater
Source Institutions
This quick demonstration (on page 11 of PDF) allows learners to understand why scientists think water ice could remain frozen in always-dark craters at the poles of the Moon.

LEGO® Chemical Reactions
Source Institutions
This activity uses LEGO® bricks to represent atoms bonding into molecules and crystals. The lesson plan is for a 2.5 hour workshop (or four 45-minute classes).
Exploring the Ocean with Robots
Source Institutions
In this activity, learners are introduced to robotic submarines called gliders. Learners make “gliders” from plastic syringes and compare these to Cartesian bottles and plastic bubbles.

Exploring Materials: Ferrofluid
Source Institutions
In this activity, learners discover that a material can act differently when it's nanometer-sized.
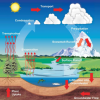
Weather Stations: Phase Change
Source Institutions
In this activity, learners observe the water cycle in action! Water vapor in a tumbler condenses on chilled aluminum foil — producing the liquid form of water familiar to us as rain and dew.

Trash Traits
Source Institutions
In this activity on page 24, learners perform experiments to examine whether or not trash can float, blow around, or wash away.

Air, It's Really There
Source Institutions
This lesson focuses on molecular motion in gases. Learners compare the mass of a basketball when it is deflated and after it has been inflated.
