Search Results
Showing results 21 to 39 of 39

Moldy Jell-O
Source Institutions
In this laboratory activity, learners design an experiment to evaluate how environmental factors influence the growth of molds.

Hot Air
Source Institutions
In this activity, learners set up an experiment to investigate the effects of hot air on the path of a laser beam.

Photosynthetic Pictures Are Worth More Than a Thousand Words
Source Institutions
This activity provides an opportunity for learners to observe and examine how carbon dioxide, water, and light produce glucose/starch through a process called photosynthesis.

Wintergreen
Source Institutions
In this outdoor, winter activity, learners find living green plants under the snow and determine the light and temperature conditions around the plants.
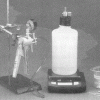
As Light as Air
Source Institutions
Learners measure a bottle full of air, and then use a vacuum pump to remove the air. When they re-weigh the bottle, learners find the mass is about 0.8g less.

Conductivity Meter
Source Institutions
In this activity, learners build a simple qualitative conductivity tester with a battery, bulb and foil.
Magnetism
Source Institutions
In this activity, learners explore the principles of magnetism by observing how various materials interact with magnets and superconductors.

Tasty Buds
Source Institutions
In this activity (1st activity on the page), learners explore their sense of taste and the structure of the tongue by taste-testing various foods.

Mussel Your Way Through Photosynthesis
Source Institutions
Using zebra mussels (Dreissena polymorpha), elodea and an indicator dye, learners study the role of light in photosynthesis.

Biological Succession in a Microecosystem
Source Institutions
This laboratory activity helps learners understand the concept of biological succession by simulating the process in a microenvironment with various protozoans.

Properties of Dust
Source Institutions
In this activity, learners carry out a scientific investigation of dust in their classroom. Learners produce an analysis on graph paper of the dust they collect over the course of a few days.

Disappearing Crystals
Source Institutions
Learners experiment with water gel crystals, or sodium polyacrylate crystals, which absorb hundreds of times their weight in water. When in pure water, the water gel crystals cannot be seen.

Infrared Investigations
Source Institutions
In this activity, learners explore how infrared technology is used by engineers to create equipment and systems for a variety of industries.

Fireworks!
Source Institutions
In this chemistry lab activity, learners model the colors of fireworks by burning metallic solutions in a flame and observing the different colors produced.

Water Underground
Source Institutions
Many people get water from a source deep underground, called groundwater.

Luminol Test
Source Institutions
Learners mix a solution containing luminol and copper with a fake blood solution. A chemical reaction between the luminol solution and fake blood (hydrogen peroxide) show learners a blue glow.

Earth's Energy Cycle: Albedo
Source Institutions
In this activity, learners experiment and observe how the color of materials that cover the Earth affects the amounts of sunlight our planet absorbs.
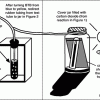
How Greenhouse Gases Absorb Heat
Source Institutions
Learners observe two model atmospheres -- one with normal atmospheric composition and another with an elevated concentration of carbon dioxide.
What Does Life Need to Live?
Source Institutions
In this astrobiology activity (on page 11 of the PDF), learners consider what organisms need in order to live (water, nutrients, and energy).
