Search Results
Showing results 21 to 40 of 321

Drying It Out
Source Institutions
In this activity, learners investigate and compare the rate of drying in different conditions.
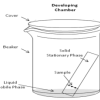
Separating with Chromatography
Source Institutions
In this experiment, learners separate different types of molecules in marker inks (using a technique called "thin layer chromatography").
Forwards and Backwards: pH and Indicators
Source Institutions
Visitors prepare six solutions combining vinegar and ammonia that range incrementally from acid (all vinegar) to base (all ammonia).

It's a Gas!
Source Institutions
In this simple activity, learners see the production of a gas, which visibly fills up a balloon placed over the neck of a bottle.

Growing Cooties
Source Institutions
In this health activity (located on page 4 of the PDF), learners will discover the importance of hand washing by observing mold growing on potatoes.

Formation of a Precipitate
Source Institutions
Learners create hard water by mixing Epsom salt and water. Then they compare what happens when soap solution is mixed with hard water and regular water.

Bready Bubble Balloon
Source Institutions
Learners discover the bubble power of living cells in this multi-hour experiment with baker's yeast. Learners make a living yeast/water solution in a bottle, and add table sugar to feed the yeast.

How Plants Grow
Source Institutions
In this biology activity (page 3 of the PDF), learners will explore how plants turn sunlight into food through a process called photosynthesis.

Traveling Through Different Liquids
Source Institutions
Learners observe and record what happens when they manipulate bottles containing a liquid (water or corn syrup) and one or more objects (screw, nail, paper clip).

Gas Production: Blow up a balloon!
Source Institutions
In this classic reaction, learners baking soda and vinegar in a soda bottle to produce carbon dioxide (CO2) gas. This gas inflates a balloon.

Which Powder is It?
Source Institutions
In this chemistry challenge, learners identify an unknown white powder by comparing it with common household powders.

Plant Power
Source Institutions
In this chemistry challenge, learners identify which plants have the enzyme "catalase" that breaks hydrogen peroxide into water and oxygen.

Who Goes There?
Source Institutions
In this outdoor, night activity, learners track nocturnal animals' footprints, droppings and other signs of their presence.

What Goes Up...
Source Institutions
In this activity about gravity (page eight of the pdf), learners will very simply explore how gravity affects objects using balls and toys.

Odors Aloft
Source Institutions
Learners smell balloons filled with different scents to guess what's inside. From this, they infer the presence and motion of scented molecules.

Oh Boy Buoyancy
Source Institutions
In this physics activity, learners will explore the concept of buoyancy, especially as it relates to density.

Kool Colors
Source Institutions
Learners investigate how temperature affects the rate of chemical reactions by observing how steel wool reacts with various types of Kool-Aid solutions at different temperatures.

Exploring How Liquids Behave
Source Institutions
Learners apply their knowledge from a previous study to identify different liquids--water, corn syrup, and vegetable oil.

Hot and Cold
Source Institutions
In this chemistry challenge, learners discover that many chemical reactions involve heat loss or gain.

The Liquid Rainbow
Source Institutions
Learners are challenged to discover the relative densities of colored liquids to create a rainbow pattern in a test tube.
