Search Results
Showing results 1 to 20 of 66

Cleaning with Dirt
Source Institutions
Learners build a filter from old soda bottles and dirt. They create polluted water, and pour it through their filter to clean it.

Surface Area
Source Institutions
In this demonstration, learners discover that nanoparticles behave differently, in part because they have a high surface area to volume ratio.
Mercury in the Environment
Source Institutions
In this environmental science lesson, learners will examine the dangers of mercury and how humans contribute to growing mercury emissions on Earth.
Trading Places: Redox Reactions
Source Institutions
Visitors add drops of copper sulfate solution onto a steel nail. They observe the nail change color from silver to brown as the copper plates onto the nail.
Currently Working: Testing Conductivity
Source Institutions
Visitors test solutions of water, sugar, salt, and hydrochloric acid and the solids salt and sugar. They clip leads from the hand generator to wires immersed in each substance.
Forwards and Backwards: pH and Indicators
Source Institutions
Visitors prepare six solutions combining vinegar and ammonia that range incrementally from acid (all vinegar) to base (all ammonia).

Clear Slime Polymer
Source Institutions
In this chemistry activity, learners use guar gum to make slime. Use this activity to introduce learners to polymers, viscosity, and colloids.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.

Pesticides and Eggshell Thinning
Source Institutions
This lab activity is about toxic substances like pesticides and their effects on biological systems. The activity starts with an introduction of how birds sequester calcium to make an egg.

Shell Shifts
Source Institutions
Ocean acidification is a big issue due to the amount of carbon dioxide humans release. CO2 in the atmosphere is absorbed into the ocean thus changing its acidity.

Get It Write
Source Institutions
In this activity, learners explore how pens have been engineered and re-engineered over time. Learners work as a team to develop a working pen out of everyday items.

Does Sunscreen Protect My DNA?
Source Institutions
In this laboratory experiment, learners explore how effectively different sunscreens protect yeast cells from damage caused by ultraviolet (UV) radiation.
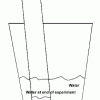
Percentage of Oxygen in the Air
Source Institutions
In this activity, learners calculate the percentage of oxygen in the atmosphere by using steel wool's ability to rust.
Yeast Balloons
Source Institutions
Visitors observe a bottle with a balloon attached around the mouth. The bottle contains a solution of yeast, sugar, and water.

3-2-1 POP!
Source Institutions
In this physics activity, learners build their own rockets out of film canisters and construction paper.

Glow Up
Source Institutions
In this activity, learners explore chemiluminescence and fluorescence. Learners examine 3 different solutions in regular light, in the dark with added bleach solution, and under a black light.

Biobarcodes: Antibodies and Nanosensors
Source Institutions
In this activity/demo, learners investigate biobarcodes, a nanomedical technology that allows for massively parallel testing that can assist with disease diagnosis.

Aerogel
Source Institutions
This activity/demo introduces learners to aerogel, a glass nanofoam. Learners discover how aerogel is made and how well it insulates as well as learn about aerogel's other unique properties.

Hanford at the Half-Life Radiation Calculator
Source Institutions
This quiz lets you estimate your annual radiation exposure.

Cook Up a Comet
Source Institutions
In this activity (on page 5 of PDF), learners use dry ice and household materials to make scientifically accurate models of comets.
