Search Results
Showing results 21 to 40 of 96
Finding the Right Crater
Source Institutions
This quick demonstration (on page 11 of PDF) allows learners to understand why scientists think water ice could remain frozen in always-dark craters at the poles of the Moon.

As The Stomach Churns
Source Institutions
In this chemistry activity, learners fill two test tubes with a solution of "artificial stomach fluid," consisting of hydrochloric acid in the same concentration as in human stomachs, some soap to cre

Exploring the Universe: Space Guess Quest
Source Institutions
Space Guess Quest is a fun game that encourages participants to identify the many types of objects in space, from human-made spacecraft to nebulas, galaxies, stars, and worlds.

Natural Buffers
Source Institutions
Learners use a universal indicator to test the amount of sodium hydroxide needed to change the pH of plain water compared with the amount needed to change the pH of gelatin.
Yeast Balloons
Source Institutions
Visitors observe a bottle with a balloon attached around the mouth. The bottle contains a solution of yeast, sugar, and water.

Common Scents
Source Institutions
Learners use a mortar and pestle to extract clove oil from cloves using denatured alcohol. They put this oil on paper, which they can take home.
Light on Other Planets
Source Institutions
In this math-based activity, learners model the intensity of light at various distances from a light source, and understand how astronomers measure the amount of sunlight that hits our planet and othe
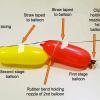
Achieving Orbit
Source Institutions
In this Engineering Design Challenge activity, learners will use balloons to investigate how a multi-stage rocket, like that used in the Interstellar Boundary Explorer (IBEX) mission, can propel a sat
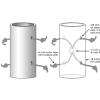
Mystery Tubes
Source Institutions
Learners investigate a pre-constructed mystery tube to determine its interior mechanism.

Flower Power
Source Institutions
In this activity, learners will experiment with changing flower colors. Learners will investigate the effects of acids and bases on flower colors.

Hot and Cold
Source Institutions
In this activity, learners explore temperature changes from chemical reactions by mixing urea with water in one flask and mixing calcium chloride with water in another flask.

Concentrate!
Source Institutions
In this investigation of reaction kinetics, learners alter the amount of iodate solution mixed with the same amount of starch solution.

Make a UV Detector
Source Institutions
In this activity, learners use tonic water to detect ultraviolet (UV) light from the Sun and explore the concept of fluorescence.

"Baseketball": A Physicist Party Trick
Source Institutions
This trick from Exploratorium physicist Paul Doherty lets you add together the bounces of two balls and send one ball flying.

Magnification vs. Resolution: Can you see the flag on the Moon?
Source Institutions
This fun and simple hands-on astronomy activity lets learners explore the difference between telescope magnification and resolution.

See the Light
Source Institutions
Learners mix a solution of luminol with hydrogen peroxide to produce a reaction that gives off blue light.

Harvesting Chemicals from a Battery
Source Institutions
In this activity, learners take apart a used zinc-carbon dry cell battery.
Dive into Design
Source Institutions
Based of the The Tech Challenge 2015, learners will engage in two mini-design challenges related to seismic engineering.

Acid Rain
Source Institutions
In this chemistry demonstration, acid rain is simulated in a petri dish.

Potato Power
Source Institutions
Learners combine hydrogen peroxide with three different forms of potato: raw chunks, ground chunks, and boiled chunks.
