Search Results
Showing results 61 to 80 of 115

Crocodiles
Source Institutions
Learners observe and compare the sizes of three toy “growing” crocodiles made from water-absorbent polymers. One is it its original state, dry, hard, and about 10cm long.

How Many Pennies?
Source Institutions
In this activity (pages 13-14), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.
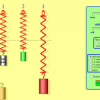
Masses & Springs
Source Institutions
In this online activity, learners use a realistic mass and spring laboratory. They hang masses from springs and adjust the spring stiffness and damping.

Plastics the Second Time Around
Source Institutions
In this activity, learners test and compare the physical properties of thermoplastic polymers. Learners compare different plastics based on their color, degree of transparency, texture, and density.

What's Your Blood Type?
Source Institutions
In this activity, learners perform a simulated blood test procedure.

Trading Places
Source Institutions
In this activity, learners discover that atoms and ions of different metals will change places.

Invisible Sunblock
Source Institutions
This is a hands-on activity exploring how nanoscale particles are used in mineral sunblocks to increase their transparency.

Home Molecular Genetics
Source Institutions
In this activity, learners extract DNA from their own cheek cells, then create a rudimentary DNA profile similar to those seen on crime scene dramas.

Crunch and Munch Lab
Source Institutions
In this activity, learners use three types of cheesy snacks--cheese balls, cheese puffs, and Cheetos--to learn about polymers.

Demonstrating An Epidemic
Source Institutions
This experiment allows learners to experience a small scale "epidemic," demonstrating the ease with which disease organisms are spread, and enables learners to determine the originator of the "epidemi

Milk Makes Me Sick: Exploration of Lactose Intolerance
Source Institutions
Why does milk make some people sick? In this activity learners explore this question and explore the chemistry of milk, and our bodies!
Ionic Bonding Puzzle Lab
Source Institutions
In this activity, learners create models of ionic compounds and observe the chemical formula of binary molecules they have created.

Dye Detective
Source Institutions
Learners analyze mixtures of dyes using filter paper chromatography. They place spots of the different dyes at the bottom of a piece of filter paper, and hang the paper to touch the surface of water.

What Does Spit Do?
Source Institutions
Some animals can swallow food whole, but humans have to chew. In this activity, learners will investigate what saliva does chemically to food before we even swallow.

Don't Throw it in the Garbage
Source Institutions
In this activity, learners discover why only some types of plastic can be recycled.
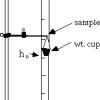
Stretching Wires
Source Institutions
In this activity, learners determine the elastic and plastic properties of different types of metal wires.

Shape Up!
Source Institutions
In this activity (pages 8-9), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Corals on Acid
Source Institutions
The objective of this inquiry-based lesson is for learners to gain an understanding of how increasing ocean acidity can affect the calcification of marine organisms.

Ready, Set, Fizz!
Source Institutions
In this activity, learners explore the chemical reaction between water and effervescent antacid tablets. This hands-on activity models how a material can act differently when it's nanometer-sized.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.
