Search Results
Showing results 1 to 20 of 58
Trading Places: Redox Reactions
Source Institutions
Visitors add drops of copper sulfate solution onto a steel nail. They observe the nail change color from silver to brown as the copper plates onto the nail.

Wheat Evolution: Sedimentation Testing
Source Institutions
In this activity (Page 30 of PDF), learners investigate the evolution of wheat by conducting sedimentation tests on different flours.
Currently Working: Testing Conductivity
Source Institutions
Visitors test solutions of water, sugar, salt, and hydrochloric acid and the solids salt and sugar. They clip leads from the hand generator to wires immersed in each substance.

Jam Jar Jet
Source Institutions
In this activity, learners create a "Jam Jar Jet" based on Francois Reynst's discovery of a pulsejet engine, which uses one opening for both air intake and exhaust.
Forwards and Backwards: pH and Indicators
Source Institutions
Visitors prepare six solutions combining vinegar and ammonia that range incrementally from acid (all vinegar) to base (all ammonia).

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.

Jem's Pykrete Challenge
Source Institutions
In this activity, learners make pykrete by freezing a mixture of water and a material like cotton wool, grass, hair, shredded paper, wood chips, or sawdust.

What Smart Metal!
Source Institutions
In this activity (pages 3-4), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.
Transformation of E. coli Using Green Fluorescent Protein
Source Institutions
In this activity related to plant biotechnology, learners transform a strain of E. coli using green fluorescent protein from a bioluminescent jellies.

Does Sunscreen Protect My DNA?
Source Institutions
In this laboratory experiment, learners explore how effectively different sunscreens protect yeast cells from damage caused by ultraviolet (UV) radiation.
Egg Osmosis
Source Institutions
Visitors observe three beakers. One beaker contains an egg immersed in vinegar. Visitors observe carbon dioxide gas escaping from the shell as the calcium carbonate reacts with the vinegar.
Yeast Balloons
Source Institutions
Visitors observe a bottle with a balloon attached around the mouth. The bottle contains a solution of yeast, sugar, and water.

Common Scents
Source Institutions
Learners use a mortar and pestle to extract clove oil from cloves using denatured alcohol. They put this oil on paper, which they can take home.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Rocket Science
Source Institutions
Learners create a small explosion by collecting hydrogen and oxygen gas together and squeezing them into a flame.

Shrinkers
Source Institutions
In this hands-on activity, learners use heat to shrink samples of polystyrene plastic (#6 recycle code). Learners compare the size and shape of the plastic pieces before and after shrinking.

Edible Glass
Source Institutions
In this activity, learners discover the principles of edible glass by making a supersaturated sugar solution.

Nanoparticle Stained Glass
Source Institutions
In this activity/demo, learners are introduced to the connection between medieval stained glass artisans and nanotechnology.
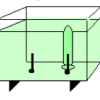
Electrolysis
Source Institutions
Using electrolysis, learners produce hydrogen gas and oxygen gas from water molecules in a solution.
Magnetism
Source Institutions
In this activity, learners explore the principles of magnetism by observing how various materials interact with magnets and superconductors.
