Search Results
Showing results 21 to 40 of 129

Nano Ice Cream
Source Institutions
In this activity/demo, learners discover how liquid nitrogen cools a creamy mixture at such a rapid rate that it precipitates super fine grained (nano) ice cream.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.

Inkjet Printer
Source Institutions
In this activity, learners investigate how inkjet printers produce tiny, precise drops of ink.

Exploring Materials: Thin Films
Source Institutions
In this activity, learners create a colorful bookmark using a super thin layer of nail polish on water. Learners discover that a thin film creates iridescent, rainbow colors.

Jem's Pykrete Challenge
Source Institutions
In this activity, learners make pykrete by freezing a mixture of water and a material like cotton wool, grass, hair, shredded paper, wood chips, or sawdust.

Bounce vs. Thud Balls
Source Institutions
Learners compare the properties of two balls that appear identical. One ball bounces, while the other ball "thuds." The “bounce” ball is made of the polymer polybutadiene (-C4H4-).
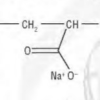
Snow Day!
Source Institutions
In this activity (on pages 4-5), learners make fake snow by adding water to the super-absorbant chemical from diapers, sodium polyacrylate.
First Impressions
Source Institutions
Learners experiment with a commercial photo-sensitive paper (Sunprint® or NaturePrint® paper). They place opaque and clear objects on the paper and expose it to bright light, observing the results.

Musical Sculpting Machine: Squeeze Play-Doh to Make Music
Source Institutions
Play-Doh is conductive! Use the semiconductive qualities of Play-Doh to make your own squeezable instrument. Pico Cricket is required.
Finding the Right Crater
Source Institutions
This quick demonstration (on page 11 of PDF) allows learners to understand why scientists think water ice could remain frozen in always-dark craters at the poles of the Moon.

Exploring Size: Tiny Ruler
Source Institutions
In this activity, learners investigate just how small a billionth of a meter is by attempting to cut a paper ruler down to a nanometer-sized sliver.

Exploring the Universe: Space Guess Quest
Source Institutions
Space Guess Quest is a fun game that encourages participants to identify the many types of objects in space, from human-made spacecraft to nebulas, galaxies, stars, and worlds.

Exploring Materials: Hydrogel
Source Institutions
In this activity, learners discover how a super-absorbing material can be used to move a straw.
Egg Osmosis
Source Institutions
Visitors observe three beakers. One beaker contains an egg immersed in vinegar. Visitors observe carbon dioxide gas escaping from the shell as the calcium carbonate reacts with the vinegar.

Forms of Carbon
Source Institutions
In this activity, educators can demonstrate how the nanoscale arrangement of atoms dramatically impacts a material’s macroscale behavior.
Yeast Balloons
Source Institutions
Visitors observe a bottle with a balloon attached around the mouth. The bottle contains a solution of yeast, sugar, and water.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

One In The Hand
Source Institutions
In this physics demonstration, learners are challenged to break a raw egg just by squeezing it. Learners will be shocked by their inability to complete the deceivingly simple challenge.

Invisible Sunblock
Source Institutions
In this activity, learners find out why some mineral sunblock rubs in clear. Learners compare nano and non-nano sunblocks and discover how particle size affects visibility.

Exploring Materials: Ferrofluid
Source Institutions
In this activity, learners discover that a material can act differently when it's nanometer-sized.
