Search Results
Showing results 1 to 20 of 28

Look-alike Liquids
Source Institutions
Learners add drops of four liquids (water, alcohol, salt water, and detergent solution) to different surfaces and observe the liquids' behavior.

How Much Water is in that Cloud?
Source Institutions
In this activity, learners working in pairs saturate a cotton ball using water drops from an eyedropper to demonstrate the high water capacity of clouds.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.
Mix and Match
Source Institutions
In this optics activity, learners explore color by examining color dots through colored water and the light of a flashlight.
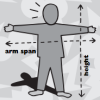
Are you a Square or a Rectangle?
Source Institutions
In this activity, learners investigate whether more people are squares or rectangles. People with similarly sized heights and arm spans are classified as squares.

Mysterious M&M's
Source Institutions
Learners place an M&M candy in water and observe what happens. The sugar-and-color coating dissolves and spreads out in a circular pattern around the M&M.

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Change in Temperature: Endothermic Reaction
Source Institutions
Learners investigate signs of a chemical reaction when they mix vinegar and baking soda. In addition to a gas being produced, learners also notice the temperature decreases.

Temperature Affects the Solubility of Gases
Source Institutions
In this activity, learners heat and cool carbonated water to find out whether temperature has an effect on how fast the dissolved gas leaves carbonated water.

Does Size Make a Difference?
Source Institutions
In this activity on page 15 of the PDF, discover how materials and physical forces behave differently at the nanoscale.

In Proportion
Source Institutions
Through this nutrition activity (page 5 of the PDF), learners will understand—and probably be surprised by—how big serving sizes of various foods should be.

Atoms and Matter (K-2)
Source Institutions
In this activity, learners explore atoms as the smallest building blocks of matter. With adult help, learners start by dividing play dough in half, over and over again.

Fingerprints
Source Institutions
In this activity, learners examine their fingerprints and learn that they can be categorized by shape, but each fingerprint is unique.

Liquid Lens
Source Institutions
In this activity, learners discover that they can create a lens from a water drop. Learners test their lens by looking at words or pictures.

Ice Cream
Source Institutions
In this chemistry activity, learners use the lowered freezing point of water to chill another mixture (ice cream) to the solid state.

Stepping Out: Hop, Skip, Jump
Source Institutions
In this activity, learners explore and experiment how we can use our bodies everyday to get from one place to another.
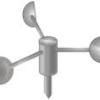
Paper Cup Anemometer
Source Institutions
In this meteorological activity, learners get to build their very own anemometer (instrument for measuring wind speed) using a paper cup.

Invisible Sunblock
Source Institutions
This is a hands-on activity exploring how nanoscale particles are used in mineral sunblocks to increase their transparency.

Push It Out
Source Institutions
In this physics related activity which requires adult supervision, learners make their own powerful water rocket and, with it, explore Newton's Third Law of Motion.

Drops on a Penny
Source Institutions
In this activity, challenge learners to predict and investigate how many water drops they can fit on one penny.
