Search Results
Showing results 1 to 17 of 17

Newton's Laws Demonstrations
Source Institutions
In this collection of demonstrations, learners explore Newton's Laws of Motion.

Separation Anxiety
Source Institutions
In this activity, learners discover the primary physical properties used to separate pure substances from mixtures.
Monster Mallows
Source Institutions
In this activity, learners explore how ordinary marshmallows expand when heated in a microwave.

Smell the Maillard Reaction
Source Institutions
In this activity, learners cook amino acids and sugar to explore the range of aromas released.

Hollandaise Sauce: Emulsion at Work
Source Institutions
In this activity, learners follow a recipe to make hollandaise sauce. Learners discover how cooks use egg yolks to blend oil and water together into a smooth mix.

Having a Gas with Cola
Source Institutions
In this activity, learners measure the amount of carbon dioxide in a carbonated drink.

Supercooled Water Drops
Source Institutions
In this activity, learners touch supercooled water drops with an ice crystal and trigger the water drops to freeze instantly.

Column Chromatography
Source Institutions
In this activity, learners separate the components of Gatorade using a home-made affinity column.
Gummy Growth
Source Institutions
In this activity related to Archimedes' Principle, learners use water displacement to compare the volume of an expanded gummy bear with a gummy bear in its original condition.
Stability of Egg White Foams
Source Institutions
In this chemistry meets cooking activity, learners compare the stability of egg white foams with various additives.

Toast a Mole!
Source Institutions
In this quick activity, learners drink Avogadro's number worth of molecules - 6.02x10^23 molecules!

Burn a Peanut
Source Institutions
In this activity, learners burn a peanut, which produces a flame that can be used to boil away water and count the calories contained in the peanut.

Light Soda
Source Institutions
In this activity, learners sublimate dry ice and then taste the carbon dioxide gas.

Condiment Diver
Source Institutions
In this hands-on activity, learners make the world's simplest Cartesian diver, using only a plastic bottle, some water, and a condiment packet.

Sweetly Balanced Equations
Source Institutions
In this (edible) activity, learners balance chemical equations using different kinds and colors of candy that represent different atoms. Learners will work in pairs and explore conservation of atoms.

Dye Like A Natural
Source Institutions
In this activity, learners stain fabrics--on purpose!
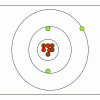
M&M® Model of the Atom: Edible Subatomic Particles
Source Institutions
In this activity, learners use colored candy to represent subatomic particles and make a model of an atom (Bohr model).
