Search Results
Showing results 1 to 20 of 60

Rollback
Source Institutions
In this physics activity (page 10 of the PDF), learners will make a can that will roll back after it has been rolled away to observe the interactions of kinetic and potential energy.

How Can Gravity Make Something Go Up?
Source Institutions
In this activity, learners use cheap, thin plastic garbage bags to quickly build a solar hot air balloon. In doing so, learners will explore why hot air rises.
Chew that Gum
Source Institutions
In this quick activity (page 1 of PDF under SciGirls Activity: Exercise and Memory), learners will investigate what happens to bubble gum when it is chewed for 5-10 minutes.
Exploring Liquids
Source Institutions
Young learners investigate and observe the properties of three liquids -- water, vegetable oil, and corn syrup. They use their senses to collect data and ask and answer questions.

Get the Porridge Just Right
Source Institutions
Learners set up three different bowls, each with a different mass of oatmeal. Learners monitor the temperature of the oatmeal and find that larger masses take longer to cool.
Thumbs Away!
Source Institutions
Learners discover how important their thumbs are by attempting various tasks without them. After tying up their thumbs, learners attempt to legibly write their names or play catch with a tennis ball.

Shell Shifts
Source Institutions
Ocean acidification is a big issue due to the amount of carbon dioxide humans release. CO2 in the atmosphere is absorbed into the ocean thus changing its acidity.
Water Filter
Source Institutions
In this engineering activity, challenge learners to invent a water filter that cleans dirty water.

Oily Ice
Source Institutions
In this activity, learners experiment with the density of ice, water, and oil. Learners will discover that the density of a liquid determines whether it will float above or sink below another liquid.

Yeast Balloons: Can biochemistry blow up a balloon?
Source Institutions
Using yeast, sugar, and water, learners create a chemical reaction which produces carbon dioxide (CO2) gas inside a 2-liter bottle. They use this gas to inflate a balloon.
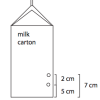
Pressing Pressure
Source Institutions
In this activity, learners compare water pressure at different depths. Learners discover that water pressure increases with depth.

What Counts in Bounce
Source Institutions
In this activity learners compare the bounciness of warm and cold racquetballs to see if temperature makes a difference in how well they bounce.

Change in Temperature: Endothermic Reaction
Source Institutions
Learners investigate signs of a chemical reaction when they mix vinegar and baking soda. In addition to a gas being produced, learners also notice the temperature decreases.
Getting Windy
Source Institutions
In this activity, learners create and understand surface currents. Learners create example surface currents and discover how landmasses affect the current.

Twirling Rope Frequency
Source Institutions
In this activity (page 1 of the PDF under SciGirls Activity: Double Dutch), learners will stand twelve feet apart swinging a rope at the slowest tempo possible while someone uses a stopwatch to record

A Dissolving Challenge
Source Institutions
In this activity, learners add objects and substances to carbonated water to discover that added objects increase the rate at which dissolved gas comes out of solution.

Wash This Way
Source Institutions
In this activity on page 4 of the PDF, learners investigate the importance of washing their hands.

Water: Clearly Unique!
Source Institutions
In this activity on page 4 of the PDF (Water in Our World), learners conduct some quick and easy tests to determine the differences between water and other liquids that look very similar to water.

Does Size Make a Difference?
Source Institutions
In this activity on page 15 of the PDF, discover how materials and physical forces behave differently at the nanoscale.

Ziptop Bag Chemistry
Source Institutions
In this chemistry activity, learners perform three chemical reactions in a sealed zip-top bag. Learners will record their observations and classify the changes as chemical or physical.
