Search Results
Showing results 1 to 20 of 46

Say Cheese!
Source Institutions
Create a chemical reaction that makes cheese! This hands-on activity demonstrates that molecules and atoms are tiny particles that make up everything around us.

A Funny Taste
Source Institutions
In this activity, learners explore the different salinities of various sources of water by taste-testing.

Art with Salt and Ice
Source Institutions
This open-ended art project allows learners to create their own colorful ice sculpture by using rock salt and food coloring on a solid block of ice.

Odors Aloft
Source Institutions
Learners smell balloons filled with different scents to guess what's inside. From this, they infer the presence and motion of scented molecules.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.

The Three Basic States (Phases) of Matter
Source Institutions
This activity was designed for blind learners, but all types of learners can explore the three states of matter by examining tactile models that illustrate the characteristics of particles in each sta

Instant Ice Cream
Source Institutions
In this activity, learners make instant ice cream without using a freezer.

3-2-1 POP!
Source Institutions
In this physics activity, learners build their own rockets out of film canisters and construction paper.
Ice Cube Painting
Source Institutions
In this activity, learners "draw" with frozen tempera paint. The ice cubes are prepared the day before by placing watered down tempera paint and popsicle sticks in ice cube trays.
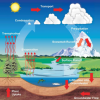
Weather Stations: Phase Change
Source Institutions
In this activity, learners observe the water cycle in action! Water vapor in a tumbler condenses on chilled aluminum foil — producing the liquid form of water familiar to us as rain and dew.

Disappearing Statues
Source Institutions
In this activity (on page 8), learners model how marble statues and buildings are affected by acid rain.

Water: Clearly Unique!
Source Institutions
In this activity on page 4 of the PDF (Water in Our World), learners conduct some quick and easy tests to determine the differences between water and other liquids that look very similar to water.

From Gas to Liquid to Solid
Source Institutions
What causes frost to form on the outside of a cold container? In this activity, learners discover that liquid water can change states and freeze to become ice.

A Mole of Gas
Source Institutions
In this two-part activity, learners use everyday materials to visualize one mole of gas or 22.4 liters of gas. The first activity involves sublimating dry ice in large garbage bag.

Cook Up a Comet
Source Institutions
In this activity (on page 5 of PDF), learners use dry ice and household materials to make scientifically accurate models of comets.

Mixing and Unmixing in the Kitchen
Source Institutions
In this chemistry investigation, learners combine common cooking substances (flour, baking powder, sugar, salt, pepper, oil, water, food coloring) to explore mixtures.

COSI Quicksand
Source Institutions
In this chemistry experiment, learners get to make a very bizarre substance using corn starch and water. Is it a solid? Is it a liquid? Or is it a different kind of substance entirely?

Personal Fan
Source Institutions
Cool off in the heat with this project! Learners use simple materials to build a fan that runs on a motor.

Atoms and Matter (K-2)
Source Institutions
In this activity, learners explore atoms as the smallest building blocks of matter. With adult help, learners start by dividing play dough in half, over and over again.

Got Gas?
Source Institutions
Create gas with a glass of water, some wire, conductors and a battery! You will be separating water (H2O) into oxygen and hydrogen.
