Search Results
Showing results 41 to 60 of 78

Oh Buoy!
Source Institutions
Learners work in pairs to design, construct, and test a device that exhibits positive, neutral, and negative buoyancy. They test a number of different objects in water to see if they sink or float.

Curious Contraptions
Source Institutions
In this engineering design activity, learners will design, test, and build a “haunting machine” to solve a Sherlockian mystery.

Balloon in a Flask
Source Institutions
Learners observe a flask with a balloon attached over the mouth and inverted inside the flask.

Layered Liquids: Chemistry You Can Drink
Source Institutions
In this chemistry activity (on page 2 of the PDF), learners make a layered drink with liquids of different densities.
Mystery Writing: Write and develop a secret message
Source Institutions
Learners write an invisible message using lemon juice on a piece of paper. They then develop the message by soaking the paper in a dilute iodine solution.

Dusting For Fingerprints
Source Institutions
In this activity, learners become detectives and use chemistry to investigate fingerprints.

What's Your Blood Type?
Source Institutions
In this activity, learners perform a simulated blood test procedure.
Color Me Blue
Source Institutions
In this activity, learners add dilute bleach solution to water that has been dyed with yellow, blue, and green food color.
Of Cabbages and Kings
Source Institutions
This lesson gives full instructions for making cabbage juice indicator, a procedure sheet for learners to record observations as they use the indicator to test materials, and extension activities to d
Floating Golf Ball
Source Institutions
Visitors observe a graduated cylinder with a golf ball floating about halfway in liquid. The bottom half of the cylinder contains a concentrated solution of salt.
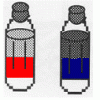
Oil and Soap
Source Institutions
Learners investigate the properties of the liquids in two bottles. One contains layers of oil and water, and one contains oil, water, and soap.

Iron in Cereal: Find iron in your food!
Source Institutions
Learners investigate an iron-fortified cereal by stirring it with a strong magnet. They discover that metallic iron is present in some cereals.

Spicy Indicator: Use turmeric to test for bases in your home
Source Institutions
This activity uses turmeric, a common spice in curry, as an indicator for acidity and basicity. Turmeric is yellow in acid and neutral substances, but turns bright red with bases.

Matter of Degree
Source Institutions
In two separate bags, learners mix water with Epsom salts and detergent.

Dye Detective
Source Institutions
Learners analyze mixtures of dyes using filter paper chromatography. They place spots of the different dyes at the bottom of a piece of filter paper, and hang the paper to touch the surface of water.

Heavy Metal
Source Institutions
In this activity (on pages 25-31 of PDF), learners soak sponges with different amounts of plaster of paris to simulate different levels of calcification in bone formation.
Good Vibrations
Source Institutions
This lesson (on pages 15-24 of PDF) explores how sound is caused by vibrating objects. It explains that we hear by feeling vibrations passing through the air.
Crystal Garden
Source Institutions
Visitors observe a tray holding a crystal-covered brick. The crystals were created by evaporation of a solution containing liquid bluing, ammonia, and salt.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
