Search Results
Showing results 1 to 20 of 216

A Funny Taste
Source Institutions
In this activity, learners explore the different salinities of various sources of water by taste-testing.

How Much Water is in that Cloud?
Source Institutions
In this activity, learners working in pairs saturate a cotton ball using water drops from an eyedropper to demonstrate the high water capacity of clouds.

Absorb & Repel
Source Institutions
In this activity, learners investigate how different materials repel or absorb water. Learners use spray bottles to explore how everyday items like sponges, cardboard, feathers, etc.

Best Bubbles
Source Institutions
In this activity, learners experiment with creating various types of bubble solutions and testing which ingredients form longer-lasting bubbles.

Exploring Materials: Liquid Crystals
Source Institutions
In this activity, learners discover that the way a material behaves on the macroscale is affected by its structure on the nanoscale.

Salt 'n Lighter
Source Institutions
In this activity, learners discover that as the salinity of water increases, the density increases as well. Learners prove this by attempting to float fresh eggs in saltwater and freshwater.

Drying It Out
Source Institutions
In this activity, learners investigate and compare the rate of drying in different conditions.
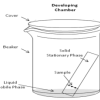
Separating with Chromatography
Source Institutions
In this experiment, learners separate different types of molecules in marker inks (using a technique called "thin layer chromatography").
Investigating Density Currents
Source Institutions
In this lab activity, learners explore how to initiate a density current. Learners measure six flasks with different concentrations of salt and water (colored blue).

Surface Tension Icebreaker
Source Institutions
This is a quick activity (located on page 2 of the PDF under Nasturtium Leaves Activity) about surface tension.

Diet Light
Source Institutions
In this quick activity, learners observe how the added sugar in a can of soda affects its density and thus, its ability to float in water.

Clear Slime Polymer
Source Institutions
In this chemistry activity, learners use guar gum to make slime. Use this activity to introduce learners to polymers, viscosity, and colloids.

Cooking with Chemistry
Source Institutions
In this activity, learners experiment with different variables in making hollandaise sauce to achieve the correct texture and consistency.

Sublime Sublimation
Source Institutions
In this activity, learners explore sublimation by conducting experiments with dry ice.

Funny Putty
Source Institutions
In this chemistry activity (page 1 of PDF SciGirls Activity: Milk Carton Boat), learners will create a blob of stretchable funny putty out of a water, borax, and glue mixture.

How Can Gravity Make Something Go Up?
Source Institutions
In this activity, learners use cheap, thin plastic garbage bags to quickly build a solar hot air balloon. In doing so, learners will explore why hot air rises.

Underwater Fireworks
Source Institutions
In this activity, learners investigate diffusion by creating underwater "fireworks" using food coloring, oil and water.

Floating Paper Clip
Source Institutions
In this activity, challenge learners to float a paper clip in a cup of water. Learners discover that a paper clip will sink in a cup of water, except when it is placed on a piece of paper towel.

Cat's Meow
Source Institutions
In this chemistry activity, learners are asked to form a hypothesis about the behavior of milk as household detergents act upon it.

Linear Functions: Mystery Liquids
Source Institutions
In this math lesson, learners analyze the density of liquids in order to explore linear functions.
