Search Results
Showing results 1 to 20 of 63

Tiny Tubes
Source Institutions
In this activity, learners make "totally tubular" forms of carbon. Learners use chicken wire to build macro models of carbon nanotubes.
Mercury in the Environment
Source Institutions
In this environmental science lesson, learners will examine the dangers of mercury and how humans contribute to growing mercury emissions on Earth.

Best Bubbles
Source Institutions
In this activity, learners experiment with creating various types of bubble solutions and testing which ingredients form longer-lasting bubbles.

Electroplating
Source Institutions
In this electrochemistry activity, learners will explore two examples of electroplating.
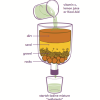
Water Clean-up
Source Institutions
This is an activity (located on page 3 of the PDF under Water Clean-up Activity) about the use of reduction agents to decontaminate ground water.

Sublime Sublimation
Source Institutions
In this activity, learners explore sublimation by conducting experiments with dry ice.

Plugged in to CO2
Source Institutions
In this activity, learners investigate various appliances and electronics, discovering how much energy each uses and how much carbon dioxide (CO2) is released to produce that energy.

Conductivity Meter
Source Institutions
In this activity, learners build a simple qualitative conductivity tester with a battery, bulb and foil.

It's a Gas, Man
Source Institutions
In this activity, learners discover if carbon dioxide has an effect on temperature.

Chemical Change
Source Institutions
In this chemistry activity, learners explore the amount of copper in a new penny. Learners use toilet bowl cleaner to hollow out the interior of a penny with zinc inside.

The Carbon Cycle and its Role in Climate Change: Activity 3
Source Institutions
In this activity, learners explore the human influences on the carbon cycle and examine how fossil fuels release carbon.

Water Wire: Electricity Flowing Through Water
Source Institutions
In this activity on page 10 of the PDF, learners detect the amount of energy that can flow through a sodium chloride electrolyte solution with a light sensor.
Salts & Solubility
Source Institutions
In this online interactive simulation, learners will add different salts to water and then watch the salts dissolve and achieve a dynamic equilibrium with solid precipitate.
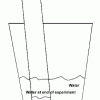
Percentage of Oxygen in the Air
Source Institutions
In this activity, learners calculate the percentage of oxygen in the atmosphere by using steel wool's ability to rust.

Flat Flashlight
Source Institutions
In this activity, learners build a tiny but powerful flashlight out of simple materials. Use this activity to introduce learners to electrical circuits and conductivity.

Hollandaise Sauce: Emulsion at Work
Source Institutions
In this activity, learners follow a recipe to make hollandaise sauce. Learners discover how cooks use egg yolks to blend oil and water together into a smooth mix.

LEGO® Chemical Reactions
Source Institutions
This activity uses LEGO® bricks to represent atoms bonding into molecules and crystals. The lesson plan is for a 2.5 hour workshop (or four 45-minute classes).

Spectroscope
Source Institutions
In this activity (posted on March 12, 2011), learners follow the steps to construct a spectroscope, a tool used to analyze light and color.
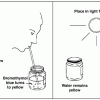
Carbon Dioxide Removal
Source Institutions
In this experiment using sprigs of Elodea, learners will observe a natural process that removes carbon dioxide (CO2) from Earth's atmosphere.
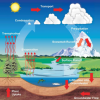
Weather Stations: Phase Change
Source Institutions
In this activity, learners observe the water cycle in action! Water vapor in a tumbler condenses on chilled aluminum foil — producing the liquid form of water familiar to us as rain and dew.
