Search Results
Showing results 21 to 40 of 62
Pollution in Our Watershed
Source Institutions
By building a simple watershed with paper and markers and then using a spray bottle to simulate precipitation, learners will understand how pollution accumulates in our water sources, especially from

School of Fish
Source Institutions
In this activity, learners will make fish cutouts that propel through the water with the help of surface tension.

Outrageous Ooze: Is It a Liquid or a Solid?
Source Institutions
This activity provides instructions for using cornstarch and water to make an ooze which has the properties of both a solid and liquid.
Current Events
Source Institutions
Learners model the ocean currents that carry hot water from the tropics to northern latitudes.
Up and Over
Source Institutions
This is an activity about Newton's First Law of Motion - a body in motion tends to stay in motion, or a body at rest tends to stay at rest unless acted upon by an outside force.

Convection Demonstration
Source Institutions
In this quick activity (located on page 2 of the PDF under GPS: Balloon Fiesta Activity), learners will see the effects of convection and understand what makes hot air balloons rise.

Turbidity
Source Institutions
This is an activity about turbidity, or the amount of sediment suspended in water.

Oh Boy Buoyancy
Source Institutions
In this physics activity, learners will explore the concept of buoyancy, especially as it relates to density.

"Can" You Stand the Pressure
Source Institutions
In this activity about states of matter, learners get to witness firsthand the awesome power of air pressure. They watch as an ordinary soda can is crushed by invisible forces.

Exploring Moisture on the Outside of a Cold Cup
Source Institutions
In this activity, learners explore the relationship between cooling water vapor and condensation. Learners investigate condensation forming on the outside of a cold cup.

Shark Sense of Smell
Source Institutions
This is an activity about our sense of smell and how it compares to sharks' super noses. Learners will create varying solutions of water and perfume.
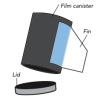
Pop Rockets
Source Institutions
In this activity, learners make film canister rocket ships. A fin pattern is glued onto the outside of the canister, and fuel (water and half an antacid tablet) is mixed inside the canister.

Changing the Density of an Object: Adding Material
Source Institutions
Learners see that a can of regular cola sinks while a can of diet cola floats. As a demonstration, bubble wrap is taped to the can of regular cola to make it float.
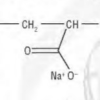
Snow Day!
Source Institutions
In this activity (on pages 4-5), learners make fake snow by adding water to the super-absorbant chemical from diapers, sodium polyacrylate.

Look-alike Liquids
Source Institutions
Learners add drops of four liquids (water, alcohol, salt water, and detergent solution) to different surfaces and observe the liquids' behavior.
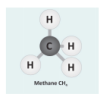
Let's Make Molecules
Source Institutions
In this activity, learners use gumdrops and toothpicks to model the composition and molecular structure of three greenhouse gases: carbon dioxide (CO2), water vapor (H2O) and methane (CH4).

Temperature Affects the Solubility of Gases
Source Institutions
In this activity, learners heat and cool carbonated water to find out whether temperature has an effect on how fast the dissolved gas leaves carbonated water.

Solving Dissolving
Source Institutions
The Sacred Cenote at Chichén Itzá is a sink hole, or well, containing groundwater. In this activity, learners create their own cenote using chalk, limestone, acids, and rain water.

Make a Lake
Source Institutions
Where rainwater goes after the rain stops? And why there are rivers and lakes in some parts of the land but not in others?
Bend a Carrot
Source Institutions
In this activity, learners investigate the process of osmosis by adding salt to a sealed bag of raw carrots and comparing it to a control.
