Search Results
Showing results 1 to 20 of 55

Surface Area
Source Institutions
In this demonstration, learners discover that nanoparticles behave differently, in part because they have a high surface area to volume ratio.

Exploring Materials: Liquid Crystals
Source Institutions
In this activity, learners discover that the way a material behaves on the macroscale is affected by its structure on the nanoscale.

Water Body Salinities I
Source Institutions
In this activity, learners investigate the different salinity levels of oceans, rivers and estuaries.

Liquid Body Armor
Source Institutions
In this activity, learners explore how nanotechnology is being used to create new types of protective fabrics.

The Electric Squeeze
Source Institutions
In this activity/demo about piezoelectricity, learners discover how some crystals produce electricity when squeezed.

Nano Ice Cream
Source Institutions
In this activity/demo, learners discover how liquid nitrogen cools a creamy mixture at such a rapid rate that it precipitates super fine grained (nano) ice cream.

Inkjet Printer
Source Institutions
In this activity, learners investigate how inkjet printers produce tiny, precise drops of ink.

Whose Fault Is It?
Source Institutions
In this seismic simulation, learners play a "who-dunnit" game to explore earthquakes.

Forgotten Genius
Source Institutions
This series of chemistry stations is designed to accompany the PBS documentary about African-American chemist "Percy Julian: Forgotten Genius." Each of the six stations features either a chemical or p
Transformation of E. coli Using Green Fluorescent Protein
Source Institutions
In this activity related to plant biotechnology, learners transform a strain of E. coli using green fluorescent protein from a bioluminescent jellies.

Does Sunscreen Protect My DNA?
Source Institutions
In this laboratory experiment, learners explore how effectively different sunscreens protect yeast cells from damage caused by ultraviolet (UV) radiation.
Finding the Right Crater
Source Institutions
This quick demonstration (on page 11 of PDF) allows learners to understand why scientists think water ice could remain frozen in always-dark craters at the poles of the Moon.

LEGO® Chemical Reactions
Source Institutions
This activity uses LEGO® bricks to represent atoms bonding into molecules and crystals. The lesson plan is for a 2.5 hour workshop (or four 45-minute classes).
Light on Other Planets
Source Institutions
In this math-based activity, learners model the intensity of light at various distances from a light source, and understand how astronomers measure the amount of sunlight that hits our planet and othe

DNA Nanotechnology
Source Institutions
In this activity, learners explore deoxyribonucleic acid (DNA), a nanoscale structure that occurs in nature.

Let's Make a Supernova!
Source Institutions
This fun and simple hands-on astronomy activity lets learners explore what happens when a star explodes.

Exploring Materials: Ferrofluid
Source Institutions
In this activity, learners discover that a material can act differently when it's nanometer-sized.
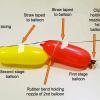
Achieving Orbit
Source Institutions
In this Engineering Design Challenge activity, learners will use balloons to investigate how a multi-stage rocket, like that used in the Interstellar Boundary Explorer (IBEX) mission, can propel a sat

Biobarcodes: Antibodies and Nanosensors
Source Institutions
In this activity/demo, learners investigate biobarcodes, a nanomedical technology that allows for massively parallel testing that can assist with disease diagnosis.

Aerogel
Source Institutions
This activity/demo introduces learners to aerogel, a glass nanofoam. Learners discover how aerogel is made and how well it insulates as well as learn about aerogel's other unique properties.
