Search Results
Showing results 1 to 18 of 18

Metal Reactions
Source Institutions
This is written as a static display, but can easily be adapted to a hands-on experiment for learners to conduct.

Wheat Evolution: Sedimentation Testing
Source Institutions
In this activity (Page 30 of PDF), learners investigate the evolution of wheat by conducting sedimentation tests on different flours.

Nano Ice Cream
Source Institutions
In this activity/demo, learners discover how liquid nitrogen cools a creamy mixture at such a rapid rate that it precipitates super fine grained (nano) ice cream.

Introduction to the New Chain Gang
Source Institutions
In this activity, learners use pop-beads to understand the characteristics and properties of polymer chains.

Fireworks!
Source Institutions
In this chemistry lab activity, learners model the colors of fireworks by burning metallic solutions in a flame and observing the different colors produced.

Why Does the Moon Have Phases?
Source Institutions
In this activity, learners use a simple 3D model to discover why the Moon has phases.

Exploring Size: Scented Balloons
Source Institutions
In this activity, learners use their sense of smell to explore the world on the nanoscale.

Standing in the Shadow of Earth
Source Institutions
This fun and simple hands-on astronomy activity demonstrates the shadow of the Earth as it rises as a dark blue shadow above the eastern horizon.

Macromodel of Microarray
Source Institutions
This is an educator-led demonstration of microarray technology using a model created from a pizza box and ping-pong balls.

Cook Up a Comet
Source Institutions
In this activity (on page 5 of PDF), learners use dry ice and household materials to make scientifically accurate models of comets.

Our Place in Our Galaxy
Source Institutions
In this fun and simple hands-on astronomy activity, learners construct a model of our place in the Milky Way Galaxy and the distribution of stars, with a quarter and some birdseed.
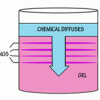
Liesegang Rings
Source Institutions
This display shows slow chemical reactions in colorful crystal formations known as Liesegang Rings. These reactions are similar to those forming the rings in agates.

Silver Crystals
Source Institutions
This is written as a static display, but can easily become a hands-on experiment for learners.

Strange Attractor: Observe Chaotic Motion
Source Institutions
In this activity, learners can observe chaotic motion. A magnet tied to a piece of string makes a pendulum, which swings over three sets of fixed magnets.

Starch Breakdown
Source Institutions
Learners use Benedict’s solution and heat to test for the presence of simple sugars in glucose, sucrose, starch, and starch combined with amylase.

Exploring Forces: Gravity
Source Institutions
In this nanoscience activity, learners discover that it's easy to pour water out of a regular-sized cup, but not out of a miniature cup.
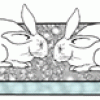
Acid Rain Eats Stone!
Source Institutions
This display shows the dangers of acid rain on buildings and other structures as two concrete bunny rabbits are disintegrated by sulfuric acid. Learners scrape chalk onto the concrete bunnies.

