Search Results
Showing results 1 to 20 of 146

Rusty Penny
Source Institutions
In this easy chemistry activity, learners submerge pennies in different liquids (water, lemon juice, vinegar, liquid hand soap, salt water, and baking soda mixed with water) to observe which best clea

Drop Shape
Source Institutions
In this activity, learners get a closer look at the shape of a drop of water and a drop of oil. Learners first drip water onto wax paper and examine the shape of separate drops from a side view.

Meteoroids and the Craters They Make
Source Institutions
In this activity, learners investigate the formation of craters. Learners will examine how the size, angle and speed of a meteorite's impact affects the properties of craters.

Magnets on the Move
Source Institutions
In this activity, learners investigate the behavior of magnets. Learners create a "wonder wand" with a magnet so they can move a skater around.

How Much Water is in that Cloud?
Source Institutions
In this activity, learners working in pairs saturate a cotton ball using water drops from an eyedropper to demonstrate the high water capacity of clouds.

Absorb & Repel
Source Institutions
In this activity, learners investigate how different materials repel or absorb water. Learners use spray bottles to explore how everyday items like sponges, cardboard, feathers, etc.

The Scoop on Habitat
Source Institutions
Some aquatic organisms live in open water, while some live in soil at the bottom of a body of water.

Colors Collide or Combine
Source Institutions
Learners place multiple M&M's in a plate of water to watch what happens as the candies dissolve.

Drying It Out
Source Institutions
In this activity, learners investigate and compare the rate of drying in different conditions.
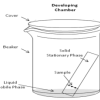
Separating with Chromatography
Source Institutions
In this experiment, learners separate different types of molecules in marker inks (using a technique called "thin layer chromatography").

It's a Gas!
Source Institutions
In this simple activity, learners see the production of a gas, which visibly fills up a balloon placed over the neck of a bottle.

Gas Production: Blow up a balloon!
Source Institutions
In this classic reaction, learners baking soda and vinegar in a soda bottle to produce carbon dioxide (CO2) gas. This gas inflates a balloon.

Who Goes There?
Source Institutions
In this outdoor, night activity, learners track nocturnal animals' footprints, droppings and other signs of their presence.

What Goes Up...
Source Institutions
In this activity about gravity (page eight of the pdf), learners will very simply explore how gravity affects objects using balls and toys.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.
Exploring Liquids
Source Institutions
Young learners investigate and observe the properties of three liquids -- water, vegetable oil, and corn syrup. They use their senses to collect data and ask and answer questions.

Vibrant Cords
Source Institutions
In this activity, learners will explore how the voice works and feel the vibrations produced by vocal cords.

Why Doesn’t the Ocean Freeze?
Source Institutions
In this activity, learners explore how salt water freezes in comparison to fresh water.
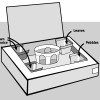
There’s No Place Like Home!
Source Institutions
In this activity, learners make their own bug boxes and test the habitat preference of selected "minibeasts" (bugs).

Jem's Pykrete Challenge
Source Institutions
In this activity, learners make pykrete by freezing a mixture of water and a material like cotton wool, grass, hair, shredded paper, wood chips, or sawdust.
