Search Results
Showing results 81 to 100 of 120

Cauldron Bubbles
Source Institutions
In this activity, learners mix up a bubbly brew and examine density. Learners explore how they can make different materials fall and rise in water using oil, water, and salt.

Nuclear Fusion
Source Institutions
This simple and engaging astronomy activity explains nuclear fusion and how radiation is generated by stars, using marshmallows as a model.

Iron in Cereal: Find iron in your food!
Source Institutions
Learners investigate an iron-fortified cereal by stirring it with a strong magnet. They discover that metallic iron is present in some cereals.

Red, White and Blue II Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the rule "likes dissolve likes" by combining three, immiscible liquids to create a colorful density column.

Erupting Fizz
Source Institutions
This is a highly visual demonstration that illustrates both the effects of density and chemical reactions.

Milk Magic
Source Institutions
In this activity, learners experiment with how dish soap and fat interact by making a colorful swirl.

Marshmallow Puff Tube
Source Institutions
In this demonstration/activity, learners observe as a regular size marshmallow is blown through a tube made from a manila file folder.
Scented Dough
Source Institutions
In this activity, learners follow a recipe to create play dough scented with fruit-flavored drink mix.

Make An Impact
Source Institutions
In this hands-on activity, learners simulate the crashing and smashing of a meteor impact using household cooking supplies.

Exponential Models: Rhinos and M&M’s ®
Source Institutions
In this math lesson, learners model exponential decay and exponential growth using M&M's, paper folding, and African rhino population data.

Natural Selection of Antibiotic Resistant Bacteria
Source Institutions
In this activity (p.3-4 of PDF), learners learn about disease transmission and antibiotic resistance.

Pea Brain!: Explorations in Estimation
Source Institutions
In this activity, learners use two different techniques to estimate how many little things fit into one bigger thing.
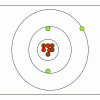
M&M® Model of the Atom: Edible Subatomic Particles
Source Institutions
In this activity, learners use colored candy to represent subatomic particles and make a model of an atom (Bohr model).

Sweet Measurements
Source Institutions
In this activity on page 3 of the PDF, learners investigate how much sugar is in a soda. Learners use sugar cubes to measure and calculate the amount of sugar in a bottle of soda.

Does Your Chewing Gum Lose Its Flavor?
Source Institutions
Each learner chews a piece of gum until it loses its flavor, and then leaves the gum to dry for several days.

Make a Green Gumball Black
Source Institutions
In this optics activity, learners use a shoebox, colored cellophane and sunlight to "change" the colors of gumballs. Learners will be surprised when the green and blue gumballs appear black!

Milli's Insulation Investigation
Source Institutions
In this activity on page 2 of the PDF, learners test different materials to find out which is the best insulator.

Soda Pop Cave
Source Institutions
In this geology activity (page 6 of the PDF), learners explore how carbonic acid can slowly dissolve limestone and form caves.

Fruits and Vegetables: Color Your Plate
Source Institutions
In this activity, learners explore healthy choices related to the foods they eat. The importance of a variety of fruits and vegetables to a healthy diet is the focus of the experience.
Gelatin Optic Fibers
Source Institutions
In this activity, learners make optical fibers out of strips of gelatin.
