Search Results
Showing results 1 to 19 of 19

Hot Stuff!: Investigation #4
Learners test two jars containing soil, one covered and one open, for changes in temperature. After placing the jars in the Sun, learners discover that the covered jar cools down more slowly.

Dripping Wet or Dry as a Bone?
Learners investigate the concept of humidity by using a dry and wet sponge as a model. They determine a model for 100% humidity, a sponge saturated with water.

Hot Stuff!: Investigation #1
Learners test two jars, one containing plain air and one containing carbon dioxide gas, to see their reactions to temperature changes.

How Can Gravity Make Something Go Up?
Source Institutions
In this activity, learners use cheap, thin plastic garbage bags to quickly build a solar hot air balloon. In doing so, learners will explore why hot air rises.

Good News: We're on the Rise!
Learners build a simple aneroid barometer to learn about changes in barometric pressure and weather forecasting. They observe their barometer and record data over a period of days.

Acid Rain Effects
Learners conduct a simple experiment to model and explore the harmful effects of acid rain (vinegar) on living (green leaf and eggshell) and non-living (paper clip) objects.
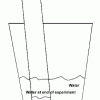
Percentage of Oxygen in the Air
Source Institutions
In this activity, learners calculate the percentage of oxygen in the atmosphere by using steel wool's ability to rust.

How can Clouds Help Keep the Air Warmer?
Source Institutions
In this activity, learners explore how air warms when it condenses water vapor or makes clouds.

Hot Stuff!: Testing for Carbon Dioxide from Our Own Breath
Learners blow into balloons and collect their breath--carbon dioxide gas (CO2). They then blow the CO2 from the balloon into a solution of acid-base indicator.

A Merry-Go-Round for Dirty Air
Learners build a model of a pollution control device--a cyclone. A cyclone works by whirling the polluted air in a circle and accumulating particles on the edges of the container.

Does Air Weigh Anything?
Source Institutions
The demonstration/experiment provides quick proof that air has mass.

If Hot Air Rises, Why is it Cold in the Mountains?
Source Institutions
This demonstration/activity helps learners understand why higher elevations are not always warm simply because "hot air rises." Learners use a tire pump to increase the pressure and temperature inside

Balloon in a Bottle
Source Institutions
In this physics activity (page 3 of the PDF), learners will see firsthand that air takes up space and has pressure by attempting to inflate a balloon inside of a bottle.

Can Crushers
Source Institutions
In this activity, learners conduct an experiment by heating an aluminum can filled with water to investigate air pressure.

Hot Stuff!: Investigation #2
Learners test two jars containing hot water, one covered with plastic and one open, for changes in temperature.

Sizing Up Temperature
Source Institutions
In this activity, learners explore Charles' Law in a syringe.

Hot Stuff!: Investigation #3
Learners test two jars of ice water, one covered and one open, for changes in temperature. After placing the jars in the sun, learners discover that the covered jar cools down more slowly.

"Boyle-ing" Water
Source Institutions
In this activity, learners explore Boyle's Law and discover that water will boil at room temperature if its pressure is lowered.

Air Pressure and Dent Pullers
Source Institutions
In this activity, learners simulate Otto von Guericke's famous Magdeburg Hemispheres experiment.
