Search Results
Showing results 161 to 180 of 285

Can Energy be Created or Destroyed?
Source Institutions
In this activity, learners explore conservation of energy by experimenting with a solar cell light device.
Paper Proteins
Source Institutions
In this activity, learners use an origami template to design eight amino acids. Learners configure the amino acids to form a protein. Use this activity to introduce proteins and amino acids.

Floating Head Cup
Source Institutions
In this activity, learners watch a figure "magically" float up through the air.

How does the Atmosphere keep the Earth Warmer?
Source Institutions
In this activity, learners simulate the energy transfer between the earth and space by using the light from a desk lamp desk lamp with an incandescent bulb and a stack of glass plates.

What is in the Water?
Source Institutions
In this activity, learners use open inquiry to learn about the process of science as well as gain experience regarding the Law of Conservation of Mass, dissolution, and density.

Let's Look at Water & the Scientific Method
Source Institutions
This activity has learners observe water and compare it to other liquids.

Rutherford Roller
Source Institutions
In this activity, learners make a black box device that serves as an excellent analogy to Rutherford's famous experiment in which he deduced the existence of the atomic nucleus.

The Squeeze is On
Source Institutions
Each group of learners uses everyday materials, such as paper, toothpicks, and tape to design and construct a structure intended to support the weight of a cinder block for 30 seconds.

Spinning Illusions
Source Institutions
In this activity, learners construct three optical illusion toys to examine how our brains play tricks on what we see.

Mirror Image
Source Institutions
In this activity, learners investigate procedural memory.
Trash Talkin'
In this activity, learners collect, categorize, weigh and analyze classroom trash and discuss ways that engineers have helped to reduce solid waste.
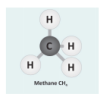
Let's Make Molecules
Source Institutions
In this activity, learners use gumdrops and toothpicks to model the composition and molecular structure of three greenhouse gases: carbon dioxide (CO2), water vapor (H2O) and methane (CH4).

Size and Scale: Probing and Predicting
Source Institutions
In this quick activity about predicting (located on page 2 of the PDF under Where's Nano?

Dusting For Fingerprints
Source Institutions
In this activity, learners become detectives and use chemistry to investigate fingerprints.

House Warming
Source Institutions
In this physical sciences activity, learners explore how passive solar design increases energy efficiency.

Breaking Point
Source Institutions
In this activity, learners build penetrometers to test leaf toughness. Biologists measure leaf toughness to study the feeding preferences of insects and bugs.

Luminescence
Source Institutions
In this two-part activity about luminescence, learners explore the chemistry that happens inside glow sticks and other light producing reactions.

Where Does the Wind Blow?
Source Institutions
In this activity, learners investigate wind by comparing the force of wind in different locations. Learners build wind-o-meters out of wooden sticks and strips of paper.

Curious Crystals
Source Institutions
Learners carefully look at four known household crystals.
Color Me Blue
Source Institutions
In this activity, learners add dilute bleach solution to water that has been dyed with yellow, blue, and green food color.
