Search Results
Showing results 1 to 20 of 232

Fizzy Nano Challenge
Source Institutions
This lesson focuses on how materials behave differently as their surface area increases.

Hot Stuff!: Investigation #4
Learners test two jars containing soil, one covered and one open, for changes in temperature. After placing the jars in the Sun, learners discover that the covered jar cools down more slowly.

Dripping Wet or Dry as a Bone?
Learners investigate the concept of humidity by using a dry and wet sponge as a model. They determine a model for 100% humidity, a sponge saturated with water.

Rusty Penny
Source Institutions
In this easy chemistry activity, learners submerge pennies in different liquids (water, lemon juice, vinegar, liquid hand soap, salt water, and baking soda mixed with water) to observe which best clea

Meteoroids and the Craters They Make
Source Institutions
In this activity, learners investigate the formation of craters. Learners will examine how the size, angle and speed of a meteorite's impact affects the properties of craters.

Hot Stuff!: Investigation #1
Learners test two jars, one containing plain air and one containing carbon dioxide gas, to see their reactions to temperature changes.

Batter Up!
Source Institutions
This activity (on page 3 of the PDF under GPS: Baseball Activity) is a full inquiry investigation into how "bounciness" relates to the distance a ball will fly when hit off a batting tee.

Measuring Your Breathing Frequency at Rest
Source Institutions
In this activity about the brain and sleep (on page 138 of the PDF), learners measure their resting breathing rates. Learners will discover that breathing frequencies vary amongst individuals.

How Much Water is in that Cloud?
Source Institutions
In this activity, learners working in pairs saturate a cotton ball using water drops from an eyedropper to demonstrate the high water capacity of clouds.

Best Bubbles
Source Institutions
In this activity, learners experiment with creating various types of bubble solutions and testing which ingredients form longer-lasting bubbles.

Statistics: Wet Heads
Source Institutions
In this math lesson, learners learn how to construct stem and leaf plots. Learners first estimate the number of drops of water that will fit on the head of a penny.

Drying It Out
Source Institutions
In this activity, learners investigate and compare the rate of drying in different conditions.
Investigating Density Currents
Source Institutions
In this lab activity, learners explore how to initiate a density current. Learners measure six flasks with different concentrations of salt and water (colored blue).
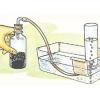
Pneumatic Trough
Source Institutions
In this activity, learners build a "pneumatic trough," a laboratory apparatus used for collecting pure gas samples over water.
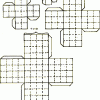
Modeling Limits to Cell Size
Source Institutions
This investigation provides learners with a hands-on activity that simulates the changing relationship of surface areas-to-volume for a growing cell.

Ball Bounce Experiment
Source Institutions
In this activity, learners investigate the properties of different types of balls.

Plot the Dot: A Graphical Approach to Density
Source Institutions
In this activity, learners work in groups to determine the mass and volume of four samples: glass marbles, steel washers or nuts, pieces of pine wood, and pieces of PVC pipe.

Surface Area and Soda Geysers
Source Institutions
This is an activity (located on page 4 of the PDF under Surface Area Activity) about surface area and reactivity.

Sublime Sublimation
Source Institutions
In this activity, learners explore sublimation by conducting experiments with dry ice.

Plugged in to CO2
Source Institutions
In this activity, learners investigate various appliances and electronics, discovering how much energy each uses and how much carbon dioxide (CO2) is released to produce that energy.
