Search Results
Showing results 1 to 20 of 36

Make Your Own Sculpture Dough
Source Institutions
In this activity on page 7 of the PDF, learners follow a recipe to make a dough similar to the clay artists use to make sculptures.

Find the Fizz: Discover the Secret of Baking Powder
Source Institutions
In this activity on page 4 of the PDF (Get Cooking With Chemistry), learners investigate ingredients that combine to produce gas bubbles.

Exploring Baking Powder
Source Institutions
In this activity, learners examine baking powder, a combination of three powders: baking soda, cream of tartar, and cornstarch.

It's a Gas!
Source Institutions
In this simple activity, learners see the production of a gas, which visibly fills up a balloon placed over the neck of a bottle.

Formation of a Precipitate
Source Institutions
Learners create hard water by mixing Epsom salt and water. Then they compare what happens when soap solution is mixed with hard water and regular water.

Universal Indicator Rainbow Trout
Source Institutions
In this activity on page 2 of the PDF, learners discover how color changes can help scientists distinguish between acids and bases.

Mystery Powders
Source Institutions
In this activity on page 2 of the PDF (Get Cooking With Chemistry), learners conduct chemical tests on certain powders used in cooking.

Bubble Trouble
Source Institutions
In this activity on page 15 of the PDF, learners measure the amount of bubbles that they make using a detergent.

Color Changes with Acids and Bases
Source Institutions
Learners mix a variety of substances with red cabbage juice. The juice changes color to indicate whether each substance is an acid or a base.

Change in Temperature: Endothermic Reaction
Source Institutions
Learners investigate signs of a chemical reaction when they mix vinegar and baking soda. In addition to a gas being produced, learners also notice the temperature decreases.

Disappearing Statues
Source Institutions
In this activity (on page 8), learners model how marble statues and buildings are affected by acid rain.

Neutralizing Acids and Bases
Source Institutions
Learners use their knowledge of color changes with red cabbage indicator to neutralize an acidic solution with a base and then neutralize a basic solution with an acid.

Wash This Way
Source Institutions
In this activity on page 4 of the PDF, learners investigate the importance of washing their hands.

New Sense about Cents
Source Institutions
In this activity on page 6 of the PDF (Chemistry—It’s Elemental), learners explore some of the properties of copper using a few common household ingredients.

Polishing Pennies
Source Institutions
In this experiment, learners try different liquids to see which ones clean pennies best. Liquids to try include water, lemon juice, cola, vinegar, and dishwashing detergent.
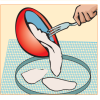
Making Recycled Paper
Source Institutions
In this activity on page 11 of the PDF, learners follow simple steps to recycle old newspaper into new paper.
Urine the Know
Source Institutions
In this activity on page 5 of the PDF, learners compare water with artificial urine to see how urinalysis works. Learners use urinalysis test strips to test for glucose and protein in the fake urine.

A Feast for Yeast
Source Institutions
In this activity on page 6 of the PDF (Get Cooking With Chemistry), learners investigate yeast. Learners prepare an experiment to observe what yeast cells like to eat.
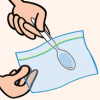
Glitter Slime
Source Institutions
In this activity on page 8 of the PDF, learners make a slimy substance very similar to mucus, and sprinkle it with glitter to imitate the way that allergens are trapped.
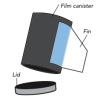
Pop Rockets
Source Institutions
In this activity, learners make film canister rocket ships. A fin pattern is glued onto the outside of the canister, and fuel (water and half an antacid tablet) is mixed inside the canister.
