Search Results
Showing results 1 to 20 of 43

Surface Area
Source Institutions
In this demonstration, learners discover that nanoparticles behave differently, in part because they have a high surface area to volume ratio.
Mercury in the Environment
Source Institutions
In this environmental science lesson, learners will examine the dangers of mercury and how humans contribute to growing mercury emissions on Earth.

Salt 'n Lighter
Source Institutions
In this activity, learners discover that as the salinity of water increases, the density increases as well. Learners prove this by attempting to float fresh eggs in saltwater and freshwater.
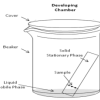
Separating with Chromatography
Source Institutions
In this experiment, learners separate different types of molecules in marker inks (using a technique called "thin layer chromatography").

Diet Light
Source Institutions
In this quick activity, learners observe how the added sugar in a can of soda affects its density and thus, its ability to float in water.

Rainbow Density Experiment
Source Institutions
In this colorful activity (page 6 of the PDF), learners will make a multicolor density column by using different concentrations of sugar solutions.

A Swell Activity with Beans
Source Institutions
In this combination chemistry and physics activity, learners explore water absorption in dried beans or peas and learn how this affects their physical properties.

Submarine: Soda Cup Lander
Source Institutions
In this activity (on page 2), learners create a submarine using a plastic cup. This is a fun way to learn about buoyancy and density.

Physics in the Kitchen: Sink or Swim Soda
Source Institutions
In the kitchen, learners can perform their own density investigation.

Having a Gas with Cola
Source Institutions
In this activity, learners measure the amount of carbon dioxide in a carbonated drink.

Forms of Carbon
Source Institutions
In this activity, educators can demonstrate how the nanoscale arrangement of atoms dramatically impacts a material’s macroscale behavior.

Rate of Solution Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the factors that increase the rate of dissolution for a solid.

Air, It's Really There
Source Institutions
This lesson focuses on molecular motion in gases. Learners compare the mass of a basketball when it is deflated and after it has been inflated.

Does Size Make a Difference?
Source Institutions
In this activity on page 15 of the PDF, discover how materials and physical forces behave differently at the nanoscale.

That's the Way the Ball Bounces: Level 1
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, size, and bounce height.

A Mole of Gas
Source Institutions
In this two-part activity, learners use everyday materials to visualize one mole of gas or 22.4 liters of gas. The first activity involves sublimating dry ice in large garbage bag.

Gassy Lava Lamp
Source Institutions
In this activity, learners use oil, water, food coloring and antacid tablets to create a bubbling lava lamp. Use this activity to introduce concepts related to density, hydrophobicity vs.
Inner Space
Source Institutions
In this activity, learners discover that there is space between molecules even in a cup "full" of water. They first fill a cup with marbles, and then add sand to fill the gaps between the marbles.

Big Things Come in Little Packages
Source Institutions
As a group, learners investigate three packages which are all the same size and shape, but have different contents. One is filled with foam, one is filled with wood, and one is filled with metal.

Pepper Scatter
Source Institutions
In this quick activity, learners break the tension that happens when water develops a "skin." Learners use water, pepper and some soap to discover the wonders of surface tension—the force that attract
