Search Results
Showing results 1 to 20 of 30
The Jelly Bean Challenge
Source Institutions
In this activity, learners explore how their sense of smell affects their taste buds. Learners taste different flavored jelly beans while holding their nose.

Colors Collide or Combine
Source Institutions
Learners place multiple M&M's in a plate of water to watch what happens as the candies dissolve.

Salt 'n Lighter
Source Institutions
In this activity, learners discover that as the salinity of water increases, the density increases as well. Learners prove this by attempting to float fresh eggs in saltwater and freshwater.

Physics in the Kitchen: Sink or Swim Soda
Source Institutions
In the kitchen, learners can perform their own density investigation.

Root Beer Float
Source Institutions
In this quick activity/demonstration about density, learners examine what happens when two cans of root beer--one diet and one regular--are placed in a large container of water.

Mysterious M&M's
Source Institutions
Learners place an M&M candy in water and observe what happens. The sugar-and-color coating dissolves and spreads out in a circular pattern around the M&M.

Rate of Solution Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the factors that increase the rate of dissolution for a solid.

One In The Hand
Source Institutions
In this physics demonstration, learners are challenged to break a raw egg just by squeezing it. Learners will be shocked by their inability to complete the deceivingly simple challenge.
Mold Terrarium: What Grows on Leftover Food?
Source Institutions
This activity shows you how to make a mold terrarium using a jar and leftover food.

Why is the Sky Blue?
Source Institutions
In this activity, learners use a flashlight, a glass of water, and some milk to examine why the sky is blue and sunsets are red.

A Stand-up Egg
Source Institutions
In this science trick, learners get an egg to stand-up on its long-axis vertical to a table's top.

Conduction Countdown
Source Institutions
In this quick SciGirls activity (page 1 of the PDF under SciGirls Activity: Doghouse Design), learners will be introduced to the concept of thermal conductivity.
Chemical Reactions in Your Mouth
Source Institutions
In this chemistry activity (page 5 of the PDF), learners will see that chewing is more than just the crushing up of food; there is actually a chemical change going on at the same time.
Fair Shares: Predict Equal Shares
Source Institutions
Use this activity to build division and number sense into any snack time or whenever there is a limited set of things to share among a group: If we deal these out, could everyone get two pieces?
Many Seeds: Estimating Hidden Seeds
Source Institutions
In this activity, learners will estimate how many seeds are in a fruit or vegetable, then count to find out. The result: mix estimation with healthy eating.
Pour Some: Measure Serving Size
Source Institutions
Make snack time into measuring time and learn to read Nutrition Facts labels. Try this when you’re using “pourable” foods, such as cereal, yoghurt, or juice.

Defining Dissolving
Source Institutions
In this introductory activity, learners discover that sugar and food coloring dissolve in water but neither dissolves in oil.
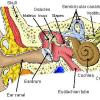
Model Eardrum
Source Institutions
In this activity (last activity on the page), learners make a model of the eardrum (also called the "tympanic membrane") and see how sound travels through the air.

Melts in Your Bag, Not in Your Hand
Source Institutions
In this activity, learners use chocolate to explore how the Sun transfers heat to the Earth through radiation.
Double or More
Source Institutions
Choose a recipe to double (or triple, quadruple, or halve). Show everyone the recipe and engage them in figuring out: How much will we need to increase the recipe to feed everyone?
